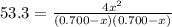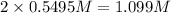Chemistry, 19.03.2020 23:27 shermoisllo3
At a certain temperature, the equilibrium constant, K c , Kc, for this reaction is 53.3. H 2 ( g ) + I 2 ( g ) − ⇀ ↽ − 2 HI ( g ) K c = 53.3 H2(g)+I2(g)↽−−⇀2HI(g)Kc=53.3 At this temperature, 0.700 mol H 2 0.700 mol H2 and 0.700 mol I 2 0.700 mol I2 were placed in a 1.00 L container to react. What concentration of HI HI is present at equilibrium?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Write skeleton equations for the following reactions c. aluminum(s)+copper(i) chloride(aq) > aluminum chloride(aq)+copper(s)
Answers: 1

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
At a certain temperature, the equilibrium constant, K c , Kc, for this reaction is 53.3. H 2 ( g ) +...
Questions



Mathematics, 09.01.2021 23:10



English, 09.01.2021 23:10


Mathematics, 09.01.2021 23:10

History, 09.01.2021 23:10

Mathematics, 09.01.2021 23:10

Mathematics, 09.01.2021 23:10

Mathematics, 09.01.2021 23:10

World Languages, 09.01.2021 23:10

Spanish, 09.01.2021 23:10


Mathematics, 09.01.2021 23:10


Mathematics, 09.01.2021 23:10


![[H_2]=\frac{0.700 mol}{1.00 L}=0.700 M](/tpl/images/0554/8381/9fcad.png)
![[I_2]=\frac{0.700 mol}{1.00 L}=0.700 M](/tpl/images/0554/8381/1f00e.png)
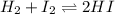
![K_c=\frac{[HI]^2}{[H_2][I_2]}](/tpl/images/0554/8381/62646.png)