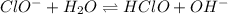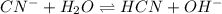Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:10
Amonoprotic acid is an acid that donates a single proton to the solution. suppose you have 0.140 g of a monoprotic acid dissolved in 35.0 ml of water. this solution is then neutralized with 14.5 ml of 0.110 m naoh. what is the molar mass of the acid?
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Chemistry, 23.06.2019 05:00
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1
You know the right answer?
Determine if each anion acts as a weak base in solution. For those anions that are basic, write an e...
Questions



Physics, 13.01.2021 21:10

Mathematics, 13.01.2021 21:10

Mathematics, 13.01.2021 21:10

Chemistry, 13.01.2021 21:10

Mathematics, 13.01.2021 21:10

Social Studies, 13.01.2021 21:10

Arts, 13.01.2021 21:10



Mathematics, 13.01.2021 21:10


German, 13.01.2021 21:10

Mathematics, 13.01.2021 21:10




English, 13.01.2021 21:10

History, 13.01.2021 21:10

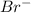 and
and 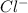 act as weak bases in solution whereas
act as weak bases in solution whereas 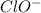 and
and 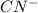 act as strong base in solution.
act as strong base in solution.