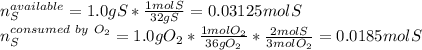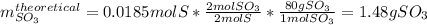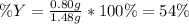Chemistry, 20.03.2020 00:28 madisonenglishp2qkow
Sulfur and oxygen react in a combination reaction to produce sulfur trioxide, an environmental pollutant: 2S(s) + 3O2(g) → 2SO3(g) In a particular experiment, the reaction of 1.0 g S with 1.0 g O2 produced 0.80 g of SO3. The % yield in this experiment is .

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
How much energy moves onto the next level, in an energy pyramid
Answers: 1

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 23.06.2019 00:20
What type of context clue you understand the meaning of quandary?
Answers: 3
You know the right answer?
Sulfur and oxygen react in a combination reaction to produce sulfur trioxide, an environmental pollu...
Questions

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Geography, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Social Studies, 16.09.2020 19:01

Geography, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Social Studies, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01

Mathematics, 16.09.2020 19:01