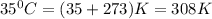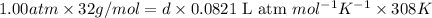Calculate the density of oxygen, O2, under each of the following conditions: STP 1.00 atm and 35.0 ∘C
Express your answers numerically in grams per liter. Enter the density at STP first and separate your answers by a comma. density at STP, density at 1 atm and 35.0 ∘C= g/L

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
Calculate the density of oxygen, O2, under each of the following conditions: STP 1.00 atm and 35.0 ∘...
Questions

History, 28.05.2020 06:57

Mathematics, 28.05.2020 06:57

Spanish, 28.05.2020 06:57


Physics, 28.05.2020 06:57


Mathematics, 28.05.2020 06:57

Mathematics, 28.05.2020 06:57



Mathematics, 28.05.2020 06:58



Mathematics, 28.05.2020 06:58

Mathematics, 28.05.2020 06:58





Geography, 28.05.2020 06:58

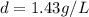 at STP,
at STP, 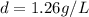 at 1 atm and
at 1 atm and 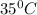
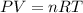
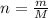
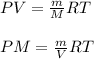
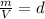 which is known as density of the gas
which is known as density of the gas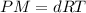
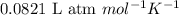
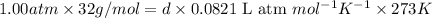
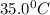 :
: