

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 23.06.2019 05:50
Which of the following is not a characteristic of s waves?
Answers: 1

Chemistry, 23.06.2019 06:30
Which of these describes how heat is transferred by convection* a. sunlight travels through space without the aid of fluids or solids. b. warm air rises and takes the heat with it, eventually, it cools and sinks c. air at the equator rises and sinks at the poles. d. air molecules touch the warm ground, heating them up *not conduction
Answers: 3
You know the right answer?
2. When 15.3 g of sodium nitrate, NaNO3,was dissolved in water in a calorimeter, the temperature fel...
Questions




Computers and Technology, 21.12.2019 06:31










Computers and Technology, 21.12.2019 06:31






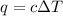
 = change in temperature =
= change in temperature = 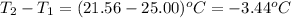
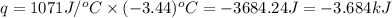

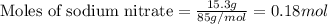
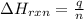
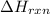 = enthalpy change of the reaction
= enthalpy change of the reaction


