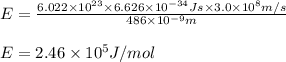Chemistry, 20.03.2020 11:30 vanessa23272
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm? (c = 3.00 × 108 m/s; h = 6.63 × 10–34 J • s; NA = 6.022 × 1023 moles–1)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 23.06.2019 08:30
Benzonitrile (c6h5cn) is reduced to two different products depending on the reducing agent used. treatment with lithium aluminum hydride followed by water forms k, which has a molecular ion in its mass spectrum at 107 and the following ir absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. treatment with a milder reducing agent forms l, which has a molecular ion in its mass spectrum at 106 and the following ir absorptions: 3086, 2850, 2820, 2736, 1703, and 1600 cm-1. l shows fragments in its mass spectrum at m/z = 105 and 77. propose structures for k and l and choose an explanation for how this could be concluded.
Answers: 3

Chemistry, 23.06.2019 10:00
Why sncl2 is solid while sncl4 is liquid at room temprature explain it in easy way
Answers: 1
You know the right answer?
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm...
Questions

English, 23.06.2021 14:20

Physics, 23.06.2021 14:30


Mathematics, 23.06.2021 14:30

History, 23.06.2021 14:30

English, 23.06.2021 14:30




English, 23.06.2021 14:30

Mathematics, 23.06.2021 14:30


English, 23.06.2021 14:30

French, 23.06.2021 14:30


Mathematics, 23.06.2021 14:30


Mathematics, 23.06.2021 14:30

English, 23.06.2021 14:30

Mathematics, 23.06.2021 14:30

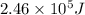
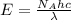
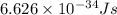
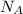 = Avogadro's number =
= Avogadro's number = 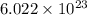
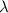 = wavelength of photon = 486 nm =
= wavelength of photon = 486 nm = 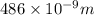 (Conversion factor:
(Conversion factor: 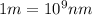 )
)