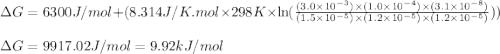Given the following: [G3P] = 1.5x10-5M; [BPG] = 3.0x10-3M ; [NAD+] = 1.2x10-5M; [NADH]=1.0x10-4 ; [HPO42-]= 1.2x10-5 M; pH = 7.5 ; DGo=6.3 kJ/mol
Glyceraldehyde3-phosphate + NAD+ + HPO42- ---> 1,3-Biphosphoglycerate + NADH + H+
Predict whether this reaction will be spontaneous.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2


Chemistry, 23.06.2019 07:40
What is the reduction potential of a hydrogen electrode that is still at standard pressure, but has ph = 5.65 , relative to the she?
Answers: 1

Chemistry, 23.06.2019 11:30
The dashed segment of the plotted experiment in the graph in the l
Answers: 3
You know the right answer?
Given the following: [G3P] = 1.5x10-5M; [BPG] = 3.0x10-3M ; [NAD+] = 1.2x10-5M; [NADH]=1.0x10-4 ; [H...
Questions

History, 16.10.2020 16:01


English, 16.10.2020 16:01

Biology, 16.10.2020 16:01

Mathematics, 16.10.2020 16:01

Mathematics, 16.10.2020 16:01



Spanish, 16.10.2020 16:01

Mathematics, 16.10.2020 16:01


Mathematics, 16.10.2020 16:01

English, 16.10.2020 16:01



Computers and Technology, 16.10.2020 16:01

English, 16.10.2020 16:01



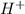 concentration, we use the equation:
concentration, we use the equation:![pH=-\log[H^+]](/tpl/images/0557/5671/cf945.png)
![7.5=-\log [H^+]](/tpl/images/0557/5671/95063.png)
![[H^+]=10^{-7.5)=3.1\times 10^{-8}M](/tpl/images/0557/5671/67b53.png)
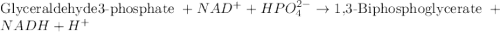
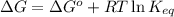
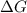 = free energy of the reaction
= free energy of the reaction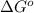 = standard Gibbs free energy = 6.3 kJ/mol = 6300 J/mol (Conversion factor: 1kJ = 1000J)
= standard Gibbs free energy = 6.3 kJ/mol = 6300 J/mol (Conversion factor: 1kJ = 1000J)![25^oC=[273+25]K=298K](/tpl/images/0557/5671/0e82f.png)
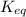 = Ratio of concentration of products and reactants =
= Ratio of concentration of products and reactants = ![\frac{[BPG][NaDH][H^+]}{[G_3P][NAD^+][HPO_4^{2-}]}](/tpl/images/0557/5671/dd6ba.png)
![[BPG]=3.0\times 10^{-3}M](/tpl/images/0557/5671/38e24.png)
![[NADH]=1.0\times 10^{-4}M](/tpl/images/0557/5671/94c0b.png)
![[H^+]=3.1\times 10^{-8}M](/tpl/images/0557/5671/2f9b6.png)
![[G_3P]=1.5\times 10^{-5}M](/tpl/images/0557/5671/aafff.png)
![[NAD^+]=1.2\times 10^{-5}M](/tpl/images/0557/5671/711eb.png)
![[HPO_4^{2-}]=1.2\times 10^{-5}M](/tpl/images/0557/5671/2ab39.png)