Chemistry, 23.03.2020 20:01 lpssprinklezlps
Suppose of iron(II) bromide is dissolved in of a aqueous solution of silver nitrate. Calculate the final molarity of bromide anion in the solution. You can assume the volume of the solution doesn't change when the iron(II) bromide is dissolved in it. Round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3
You know the right answer?
Suppose of iron(II) bromide is dissolved in of a aqueous solution of silver nitrate. Calculate the f...
Questions



History, 05.05.2020 21:03



History, 05.05.2020 21:03


Mathematics, 05.05.2020 21:03

Mathematics, 05.05.2020 21:03



English, 05.05.2020 21:03


Mathematics, 05.05.2020 21:03


History, 05.05.2020 21:03



Mathematics, 05.05.2020 21:03

Engineering, 05.05.2020 21:03

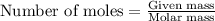
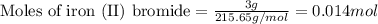
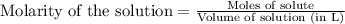 .....(1)
.....(1)
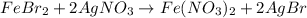
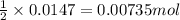 of iron (II) bromide
of iron (II) bromide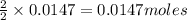 of silver bromide
of silver bromide