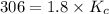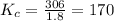Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 23:00
What is the name of the enzyme that forms at the start of transcription?
Answers: 1

Chemistry, 23.06.2019 01:30
In what way do investigations build scientific knowledge? the results of investigations lead to questions that cannot be tested. they reflect the opinions and social values of scientists, ensuring valid information. the results of investigations lead to new questions, which lead to new investigations. they are not influenced by the research of earlier scientists, so they are able to address gaps in understanding.i
Answers: 1
You know the right answer?
Carbon monoxide replaces oxygen in oxygenated hemoglobin according to the reaction: HbO2(aq) + CO(aq...
Questions

Mathematics, 13.10.2019 20:50

English, 13.10.2019 20:50



Mathematics, 13.10.2019 20:50



Mathematics, 13.10.2019 20:50

Biology, 13.10.2019 20:50

Mathematics, 13.10.2019 20:50

Biology, 13.10.2019 20:50



Mathematics, 13.10.2019 20:50



Mathematics, 13.10.2019 20:50

Mathematics, 13.10.2019 20:50


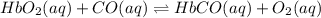
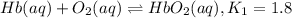
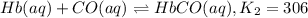
![K_1=\frac{[HbO_2]}{[Hb][O_2]}](/tpl/images/0560/2528/dcc99.png)
![[Hb]=\frac{[HbO_2]}{[K_1][O_2]}](/tpl/images/0560/2528/1a6fb.png) ..[1]
..[1]![K_2=\frac{[HbCO]}{[Hb][CO]}](/tpl/images/0560/2528/abc0a.png) ..[2]
..[2]![K_c=\frac{[HbCO][O_2]}{[HbO_2][CO]}](/tpl/images/0560/2528/bc291.png) ..[3]
..[3]![K_2=\frac{[HbCO]}{\frac{[HbO_2]}{[K_1][O_2]}\times [CO]}](/tpl/images/0560/2528/e05b3.png)
![K_2=K_1\times \frac{[HbCO][O_2]}{[HbO_2][CO]}](/tpl/images/0560/2528/e36c4.png)
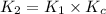 ( using [3])
( using [3])