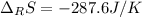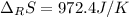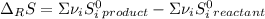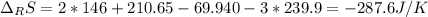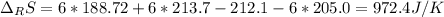Chemistry, 24.03.2020 05:55 giuliabril
Calculate ΔS°rxn (J/k) for 3NO2(g) + H2O(l)LaTeX: \longrightarrow⟶ NO(g) +2HNO3(l) C6H12O6(s) + 6O2(g) LaTeX: \longrightarrow⟶ 6H2O(g) +6CO2(g) Enter numbers to 1 decimal places. Substance or Ion S° (J/molLaTeX: \cdot⋅K) N2(g) 191.5 N2O(g) 219.7 NO(g) 210.65 NO2(g) 239.9 F2(g) 202.7 H2(g) 130.6 HNO3(l) 155.6 HNO3(aq) 146 H2O(l) 69.940 H2O(g) 188.72 C6H12O6(s) 212.1 O2(g) 205.0 CO2(g) 213.7 CO2(aq) 121 NF3(g) 260.6

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

You know the right answer?
Calculate ΔS°rxn (J/k) for 3NO2(g) + H2O(l)LaTeX: \longrightarrow⟶ NO(g) +2HNO3(l) C6H12O6(s) + 6O2(...
Questions

Mathematics, 07.10.2019 16:30

History, 07.10.2019 16:30



Computers and Technology, 07.10.2019 16:30

History, 07.10.2019 16:30

Mathematics, 07.10.2019 16:30

English, 07.10.2019 16:30

Physics, 07.10.2019 16:30




Mathematics, 07.10.2019 16:30

Social Studies, 07.10.2019 16:30




History, 07.10.2019 16:30

Social Studies, 07.10.2019 16:30