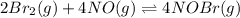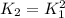Chemistry, 24.03.2020 17:51 rustalex6045
Consider the equilibrium reaction and its equilibrium constant expression. Br 2 ( g ) + 2 NO ( g ) − ⇀ ↽ − 2 NOBr ( g ) K = [ NOBr ] 2 [ Br 2 ] [ NO ] 2 For the reaction 2 Br 2 ( g ) + 4 NO ( g ) − ⇀ ↽ − 4 NOBr ( g ) select the equilibrium constant expression.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1



Chemistry, 23.06.2019 06:30
Generally, observed behavior that can be formulated into a statement, sometimes mathematical in nature, is called a(n): a. observation. b. measurement. c. theory. d. natural law. e. experiment.
Answers: 2
You know the right answer?
Consider the equilibrium reaction and its equilibrium constant expression. Br 2 ( g ) + 2 NO ( g ) −...
Questions

Spanish, 13.12.2019 10:31


Social Studies, 13.12.2019 10:31

Mathematics, 13.12.2019 10:31






Mathematics, 13.12.2019 10:31

English, 13.12.2019 10:31

Mathematics, 13.12.2019 10:31




Mathematics, 13.12.2019 10:31

Mathematics, 13.12.2019 10:31



Mathematics, 13.12.2019 10:31

![K_2=\frac{[NOBr]^4_{eq}}{[NO]^4_{eq}[Br]^2_{eq}}](/tpl/images/0560/9944/3b128.png)