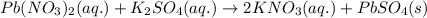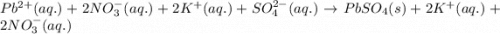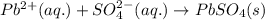Chemistry, 24.03.2020 19:38 aurorasweetpea610
Enter a net ionic equation to show the reaction of aqueous lead(II) nitrate with aqueous potassium sulfate to form solid lead(II) sulfate and aqueous potassium nitrate. Express your answer as a chemical equation. Identify all of the phases in your answer.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Embryos of different species look very similar, which shows that the organisms share a ancestor.
Answers: 1

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration
Answers: 1

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1
You know the right answer?
Enter a net ionic equation to show the reaction of aqueous lead(II) nitrate with aqueous potassium s...
Questions

English, 18.12.2020 21:50


SAT, 18.12.2020 21:50


Mathematics, 18.12.2020 21:50

Mathematics, 18.12.2020 21:50


Mathematics, 18.12.2020 21:50


History, 18.12.2020 21:50



Mathematics, 18.12.2020 21:50

Social Studies, 18.12.2020 21:50

Mathematics, 18.12.2020 21:50

Chemistry, 18.12.2020 21:50


Mathematics, 18.12.2020 21:50

Physics, 18.12.2020 21:50