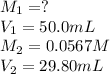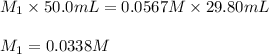Chemistry, 25.03.2020 00:28 itzdulceee
Acetic acid (CH3CO2H) is the principal component in the vinegar. What is the molarity of an acetic acid solution if a titration of 50.00 mL of the acetic acid solution requires 29.80 mL of 0.0567 M NaOH to reach the equivalence point?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 05:30
Arecipe calls for 1.2 cups of oil. how many liters of oil is this?
Answers: 2

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1
You know the right answer?
Acetic acid (CH3CO2H) is the principal component in the vinegar. What is the molarity of an acetic a...
Questions

Mathematics, 01.06.2020 20:57


Mathematics, 01.06.2020 20:57

History, 01.06.2020 20:57

Chemistry, 01.06.2020 20:57


Mathematics, 01.06.2020 20:57

Mathematics, 01.06.2020 20:57

Mathematics, 01.06.2020 20:57

Engineering, 01.06.2020 20:57



History, 01.06.2020 20:57

Mathematics, 01.06.2020 20:57

Mathematics, 01.06.2020 20:57

English, 01.06.2020 20:57



Biology, 01.06.2020 20:57

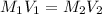
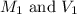 are the molarity and volume of acetic acid.
are the molarity and volume of acetic acid.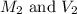 are the molarity and volume of NaOH.
are the molarity and volume of NaOH.