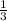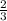Chemistry, 25.03.2020 01:21 FavvBella84
The total pressure for a mixture of N2O4 and NO2 is 1.5atm. If Kp = 7.1 at 25°C, calculate the partial pressure of each gas in the mixture. (HINT 1.5 = 2pNO2 + pN2O4) 2NO2 (g) = N2O4 (g)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1


Chemistry, 22.06.2019 22:00
If a solution contains 3 moles/liter of sodium chloride (nacl, made of sodium ions and chloride ions), what is the osmolarity of this solution
Answers: 3
You know the right answer?
The total pressure for a mixture of N2O4 and NO2 is 1.5atm. If Kp = 7.1 at 25°C, calculate the parti...
Questions



Mathematics, 20.09.2021 09:50

History, 20.09.2021 09:50




Mathematics, 20.09.2021 09:50

English, 20.09.2021 09:50




Mathematics, 20.09.2021 09:50

Social Studies, 20.09.2021 09:50






Chemistry, 20.09.2021 09:50

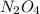 : 0.5 atm and
: 0.5 atm and 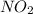 : 1 atm
: 1 atm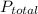 = 1.5 atm
= 1.5 atm