
Chemistry, 25.03.2020 01:57 wedderman6049
At a certain temperature, the reaction 2NO + Cl2 ⇌ 2NOCl has an equilibrium constant Kc of 45.0. A chemist creates a mixture with the following initial concentrations: [NO] = 0.10 M, [Cl2] = 0.20 M, and [NOCl] = 0.30 M. What will happen as the reaction begins?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1
You know the right answer?
At a certain temperature, the reaction 2NO + Cl2 ⇌ 2NOCl has an equilibrium constant Kc of 45.0. A c...
Questions

Mathematics, 06.05.2020 00:32






Geography, 06.05.2020 00:32









Mathematics, 06.05.2020 00:32

History, 06.05.2020 00:32



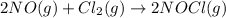 has an equilibrium constant Kc of 45.0. A chemist creates a mixture with the following initial concentrations: [NO] = 0.10 M, [Cl₂] = 0.20 M, and [NOCl] = 0.30 M. What will happen as the reaction begins?
has an equilibrium constant Kc of 45.0. A chemist creates a mixture with the following initial concentrations: [NO] = 0.10 M, [Cl₂] = 0.20 M, and [NOCl] = 0.30 M. What will happen as the reaction begins?![Q=\frac{[NOCl]^2}{[NO]^2[Cl_2]}](/tpl/images/0562/1779/afbe9.png)
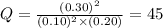
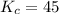
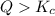 that means product > reactant. So, the reaction is reactant favored.
that means product > reactant. So, the reaction is reactant favored.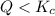 that means reactant > product. So, the reaction is product favored.
that means reactant > product. So, the reaction is product favored.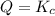 that means product = reactant. So, the reaction is in equilibrium.
that means product = reactant. So, the reaction is in equilibrium.

