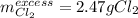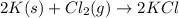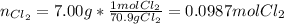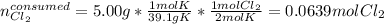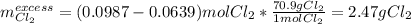Chemistry, 25.03.2020 04:55 arizola757
Potassium chloride is used as a substitute for sodium chloride for individuals with high blood pressure. Identify the limiting reactant and determine the mass of the excess reactant remaining when 7.00 g of chlorine gas reacts with 5.00 g of potassium to form potassium chloride.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
You know the right answer?
Potassium chloride is used as a substitute for sodium chloride for individuals with high blood press...
Questions

Mathematics, 09.06.2021 18:20


Physics, 09.06.2021 18:20






Biology, 09.06.2021 18:20



Mathematics, 09.06.2021 18:20


Mathematics, 09.06.2021 18:20

Mathematics, 09.06.2021 18:20

Mathematics, 09.06.2021 18:20

History, 09.06.2021 18:20

Mathematics, 09.06.2021 18:20

Mathematics, 09.06.2021 18:20