
Chemistry, 26.03.2020 04:28 jeffffffff
3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution's temperature increases by 19.2°C. Calculate ∆H for the dissolution of the unknown solid. (The specific heat of the solution is 4.18 J/g・°C and the density of the solution is 1.20 g/mL).

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 04:20
Neils bohr believed that electrons orbited the nucleus in different energy levels, based on strong support from
Answers: 1

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3
You know the right answer?
3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution'...
Questions


English, 01.07.2019 00:00


Mathematics, 01.07.2019 00:00

English, 01.07.2019 00:00

English, 01.07.2019 00:00



Biology, 01.07.2019 00:00

Mathematics, 01.07.2019 00:00


Chemistry, 01.07.2019 00:00


Mathematics, 01.07.2019 00:00

Mathematics, 01.07.2019 00:00





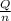 =
= 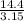 = 4.6 kj/mole
= 4.6 kj/mole

