
Chemistry, 26.03.2020 21:12 torquishag
Consider the following reaction in which all reactants and products are gases. 1.00 mol of A and 2.00 mol of B are placed in a 5.0-liter container. After equilibrium has been established, 0.50 mol of D is present in the container. Calculate the equilibrium constant, Kc, for the reaction.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
A32 year old immigrant from a patriarchal country is giving birth. as she is delivering the baby, she tearfully confesses to her doctor that this is her 4th child and she simply cannot handle any more children. she tells the doctor that her husband refuses to use contraception or allow her to, and she begs her doctor to tie her tubes and not tell her husband. the doctor complies. was hipaa violated? why or why not?
Answers: 3

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3


Chemistry, 22.06.2019 18:30
Which rate indicates the number of children that would be born per woman if she were to live to the end of her child bearing years
Answers: 2
You know the right answer?
Consider the following reaction in which all reactants and products are gases. 1.00 mol of A and 2.0...
Questions

History, 31.08.2021 06:00

Physics, 31.08.2021 06:10



Chemistry, 31.08.2021 06:10

Mathematics, 31.08.2021 06:10

Mathematics, 31.08.2021 06:10

Health, 31.08.2021 06:10


Social Studies, 31.08.2021 06:10

Mathematics, 31.08.2021 06:10

Mathematics, 31.08.2021 06:10

Mathematics, 31.08.2021 06:10


Mathematics, 31.08.2021 06:10


Mathematics, 31.08.2021 06:10

History, 31.08.2021 06:10


![K_{c} = \frac{[D]}{[A][B]}](/tpl/images/0565/8107/95178.png) =
= 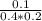 =1.25
=1.25

