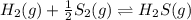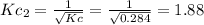Chemistry, 26.03.2020 21:03 estefaniapenalo
When heated, hydrogen sulfide gas decomposes according to the equation: 2H2S(g) → 2H2(g) + 2S2(g) A 6.75 gram sample of H2S(g) is introduced into an evacuated rigid 0.75 L container. The sealed container is heated to 283 K and 6.42 x 10 ^–2 mol of S2 gas is present at equilibrium.
a. Calculate the equilibrium concentration, in mol/L, of the H2(g) in the container at 283 K.
b. Calculate the equilibrium concentration, in mol/L, of the H2S(g) in the container at 283 K.
c. Calculate the value of the equilibrium constant, Kc, for the decomposition reaction at 283 K.
d. Calculate the partial pressure of S2(g) in atm in the container at equilibrium at 283 K.
e. Calculate the value of the equilibrium constant, Kc, for the reaction
H2(g) + 1/2 S2(g) → H2S (g) at 283 K.

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 00:10
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3

Chemistry, 23.06.2019 04:10
In an experiment, 45g of silicon tetrachloride are treated with 45ml of water. what is the theoretical yield in grams of hcl
Answers: 3

Chemistry, 23.06.2019 07:10
Which one of the following is an oxidation-reduction reaction? naoh + hno3 --> h2o + kno3 naoh + hno3 --> h2o + kno3 so3 + h2o --> h2so4 cacl2 + na2co3 --> caco3 + 2 nacl ch4 + 2 o2 --> co2 + 2 h2o al2(so4)3 + 6 koh --> 2 al(oh)3 + 3 k2so4
Answers: 3

Chemistry, 23.06.2019 13:00
How does the kinetic energy of a substance's particle in the solid phase compare to their kinetic enegy in the liquid phase?
Answers: 1
You know the right answer?
When heated, hydrogen sulfide gas decomposes according to the equation: 2H2S(g) → 2H2(g) + 2S2(g) A...
Questions


Physics, 08.10.2021 22:00



Mathematics, 08.10.2021 22:00


History, 08.10.2021 22:00

Mathematics, 08.10.2021 22:00

English, 08.10.2021 22:00



Biology, 08.10.2021 22:00

Mathematics, 08.10.2021 22:10

Arts, 08.10.2021 22:10

Social Studies, 08.10.2021 22:10

Computers and Technology, 08.10.2021 22:10

Mathematics, 08.10.2021 22:10



Mathematics, 08.10.2021 22:10


 due to stoichiometry and the reaction extent, turns out:
due to stoichiometry and the reaction extent, turns out:![K=\frac{[H_2]_{eq}^2[S_2]_{eq}}{[H_2S]_{eq}^2}](/tpl/images/0565/7677/a5ff5.png)
![[H_2S]_0=\frac{6.75g/(34g/mol)}{0.75L} =0.265M](/tpl/images/0565/7677/26ae9.png)
![[S_2]_{eq}=x=\frac{6.42x10^{-2}mol}{0.75L}=0.0856M](/tpl/images/0565/7677/c4de0.png)
![[H_2]_{eq}=2x=2*0.0856M=0.171M](/tpl/images/0565/7677/0a782.png)
![[H_2S]_{eq}=0.265M-2x=0.265M-2*0.0856M=0.0938M](/tpl/images/0565/7677/de91b.png)
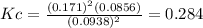
![p_{S_2}=[S_2]RT= 0.0856\frac{mol}{L}*0.082\frac{atm*L}{mol*K}*283K=1.99atm](/tpl/images/0565/7677/136fa.png)