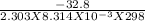Chemistry, 26.03.2020 23:00 daphnevlogs11
The Haber process for the production of ammonia is the main industrial process of producing ammonia today. Prior to developing this process, ammonia was difficult to produce on an industrial scale. The reaction for the Haber process is: N2(g) + 3 H2(g) ⇌ 2 NH3(g). Using the following values, determine the equilibrium constant for this reaction at 25oC. Substance ΔGfo(kJ/mol) N2 0 H2 0 NH3 -16.4

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1


Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1
You know the right answer?
The Haber process for the production of ammonia is the main industrial process of producing ammonia...
Questions







Mathematics, 10.05.2021 22:20




Mathematics, 10.05.2021 22:20





Mathematics, 10.05.2021 22:20

Biology, 10.05.2021 22:20

Social Studies, 10.05.2021 22:20


English, 10.05.2021 22:20