Chemistry, 26.03.2020 23:42 nadarius2017
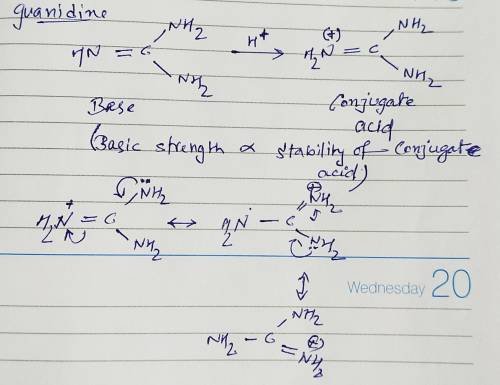
Guanidine is a stronger base than the typical amine. The increased basicity can be explained by drawing the resonance structures of the protonated guanidine. The protonated guanidine (A) has been drawn for you. Draw major resonance structures, one each in boxes B and C, and one minor resonance structure in Box D. Be sure to include the formal charge, lone pairs, and hydrogens on nitrogen for structures B, C, and D.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:30
If a 60-g object has a volume of 30 cm3, what is its density? 2 g/cm3 0.5 cm3/g 1800 g * cm3 none of the above
Answers: 3

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 21.06.2019 21:00
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1
You know the right answer?
Guanidine is a stronger base than the typical amine. The increased basicity can be explained by draw...
Questions


Mathematics, 23.03.2021 20:20

Mathematics, 23.03.2021 20:20

History, 23.03.2021 20:20


Physics, 23.03.2021 20:20

Mathematics, 23.03.2021 20:20

Mathematics, 23.03.2021 20:20


Mathematics, 23.03.2021 20:20

Chemistry, 23.03.2021 20:20

Arts, 23.03.2021 20:20


Social Studies, 23.03.2021 20:20

English, 23.03.2021 20:20




Mathematics, 23.03.2021 20:20