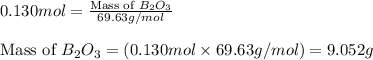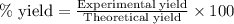Chemistry, 29.03.2020 21:45 demarpratt1270
Find percent yield:
The mass of B2O3 produced by the reaction of 4.00 g of B5H9, and 10.00 g of
O2 is 8.32 g. What is the percent yield?
2 B3H9 + 12 O2 => 5 B2O3 +9 H2O
87.2
92.8
91.8
75.5
74.5

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1

Chemistry, 23.06.2019 05:00
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3
You know the right answer?
Find percent yield:
The mass of B2O3 produced by the reaction of 4.00 g of B5H9, and 10....
The mass of B2O3 produced by the reaction of 4.00 g of B5H9, and 10....
Questions

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

English, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

English, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Physics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

History, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

Mathematics, 16.09.2020 18:01

 .....(1)
.....(1)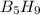 :
: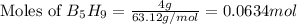
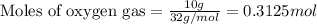
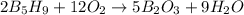
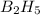
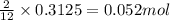 of
of 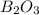
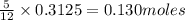 of water
of water