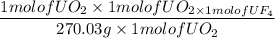Using the unbalanced reaction below:
UO2(s) + HF(aq) > UF4 + H2O(l)
How m...

Chemistry, 30.03.2020 01:38 ayoismeisalex
Using the unbalanced reaction below:
UO2(s) + HF(aq) > UF4 + H2O(l)
How many grams of UF4 would we anticipate to get from 1.00 g of UO2 and an excess of HF

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1

Chemistry, 23.06.2019 07:30
Which statement explains which thermometer is more appropriate to measure the temperature of a liquid at 43.6 degrees celsius a) thermometer a, because it measures temperature more accurately than thermometer b b) thermometer b, because it measures temperature more accurately than thermometer a c) thermometer a, because it measures temperature more precisely than thermometer b d) thermometer b, because it measures temperature more precisely than thermometer a
Answers: 2

Chemistry, 23.06.2019 19:30
Type the correct answer in the box. tungsten carbide is becoming popular for making jewelry. a chemist is examining a sample of tungsten carbide (chemical formula wc). what is the percent composition of tungsten in this compound? round your answer to the nearest whole percent. the percent composition of tungsten is what %?
Answers: 1

Chemistry, 24.06.2019 03:30
In a synthesis reaction, one reactant contains 256 j of chemical energy, and one reactant contains 328 j of chemical energy. the product contains 407 j of chemical energy. because energy is conserved, what energy change occurs during the reaction? a. 177 j of energy is released. b. 335 j of energy is absorbed. c. 177 j of energy is absorbed. d. 335 j of energy is released.
Answers: 2
You know the right answer?
Questions




Biology, 08.07.2019 08:00

Mathematics, 08.07.2019 08:00

Mathematics, 08.07.2019 08:00




Mathematics, 08.07.2019 08:00

English, 08.07.2019 08:00

Geography, 08.07.2019 08:00



Mathematics, 08.07.2019 08:00


Mathematics, 08.07.2019 08:00

Mathematics, 08.07.2019 08:00


History, 08.07.2019 08:00