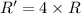Chemistry, 30.03.2020 17:31 angelolucero146
) Given the following rate law, how does the rate of reaction change if the concentration of Y is doubled? Rate = k [X][Y]2 A) The rate of reaction will increase by a factor of 2. B) The rate of reaction will increase by a factor of 5. C) The rate of reaction will increase by a factor of 4. D) The rate of reaction will decrease by a factor of 2. E) The rate of reaction will remain unchanged.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
What problem would a person have if the nucleic acid in one of his or her cells were damaged?
Answers: 2

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2
You know the right answer?
) Given the following rate law, how does the rate of reaction change if the concentration of Y is do...
Questions

Mathematics, 26.06.2020 16:01

Mathematics, 26.06.2020 16:01

Mathematics, 26.06.2020 16:01

History, 26.06.2020 16:01



Mathematics, 26.06.2020 16:01







Mathematics, 26.06.2020 16:01

Social Studies, 26.06.2020 16:01



Mathematics, 26.06.2020 16:01


![Rate(R) = k [X][Y]^2](/tpl/images/0570/5331/25584.png)
![R'=k[X][2Y]^2](/tpl/images/0570/5331/8ce13.png)
![R'=k[X]4[Y]^2](/tpl/images/0570/5331/f56ad.png)
![R'=4k[X][Y]^2](/tpl/images/0570/5331/59783.png)