
Chemistry, 30.03.2020 17:30 johnsonkia873
Write chemical equations and corresponding equilibrium expressions for each of the two ionization steps of carbonic acid. Part A Write chemical equations for first ionization step of carbonic acid. Express your answer as a chemical equation. Identify all of the phases in your answer. nothing Request Answer Part B Complete previous part(s) Part C Write chemical equations for second ionization step of carbonic acid. Express your answer as a chemical equation. Identify all of the phases in your answer. nothing

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1

Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 1
You know the right answer?
Write chemical equations and corresponding equilibrium expressions for each of the two ionization st...
Questions


Mathematics, 10.04.2020 20:10




Mathematics, 10.04.2020 20:10


Mathematics, 10.04.2020 20:10



Chemistry, 10.04.2020 20:10


Mathematics, 10.04.2020 20:10


Mathematics, 10.04.2020 20:10


Social Studies, 10.04.2020 20:10

English, 10.04.2020 20:10


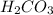 . It is a diprotic weak acid which means that it will release two hydrogen ions when dissolved in water
. It is a diprotic weak acid which means that it will release two hydrogen ions when dissolved in water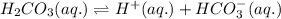
![Ka_1=\frac{[H^+][HCO_3^{-}]}{[H_2CO_3]}](/tpl/images/0570/5262/fb18f.png)
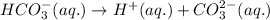
![Ka_2=\frac{[H^+][CO_3^{2-}]}{[HCO_3^-]}](/tpl/images/0570/5262/f465d.png)


