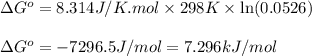Chemistry, 30.03.2020 19:05 jojoangelique13
Phosphoglucoisomerase interconverts glucose 6‑phosphate to, and from, glucose 1‑phosphate. glucose 6 - phosphate phosphoglucoisomerase ⇌ glucose 1 - phosphate glucose 6-phosphate⇌ 1-phosphate After mixing equal amounts of the two molecules, the solution achieved equilibrium at 25 ∘ C . 25 ∘C. The concentration at equilibrium of glucose 1‑phosphate is 0.01 M, 0.01 M, and the concentration at equilibrium of glucose 6‑phosphate is 0.19 M. 0.19 M. Calculate the equilibrium constant, K eq , Keq, and the standard free energy change, Δ G ∘ , ΔG∘, of the reaction mixture.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
1. explain hydrogen peroxide, h 2 o 2 properties and decomposition reaction. 2. describe how each of the following natural cycles plays a part in earth’s climate system. (a) the water cycle (b) the carbon cycle
Answers: 1

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1

Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2
You know the right answer?
Phosphoglucoisomerase interconverts glucose 6‑phosphate to, and from, glucose 1‑phosphate. glucose 6...
Questions

Chemistry, 12.02.2021 21:00

Mathematics, 12.02.2021 21:00


Spanish, 12.02.2021 21:00


Social Studies, 12.02.2021 21:00

Mathematics, 12.02.2021 21:00



Mathematics, 12.02.2021 21:00

Mathematics, 12.02.2021 21:00


Mathematics, 12.02.2021 21:00

Mathematics, 12.02.2021 21:00



Biology, 12.02.2021 21:00

Health, 12.02.2021 21:00

Mathematics, 12.02.2021 21:00

Social Studies, 12.02.2021 21:00

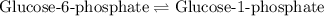
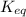 for above equation follows:
for above equation follows:![K_{eq}=\frac{[\text{Glucose-1-phosphate}]}{[\text{Glucose-6-phosphate}]}](/tpl/images/0570/7623/fc6f1.png)
![[\text{Glucose-1-phosphate}]=0.01M](/tpl/images/0570/7623/c031f.png)
![[\text{Glucose-6-phosphate}]=0.19M](/tpl/images/0570/7623/d838e.png)
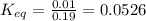
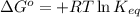
 = Standard Gibbs free energy
= Standard Gibbs free energy![25^oC=[25+273]K=298K](/tpl/images/0570/7623/df1f6.png)