Chemistry, 30.03.2020 20:35 fireemblam101ovu1gt
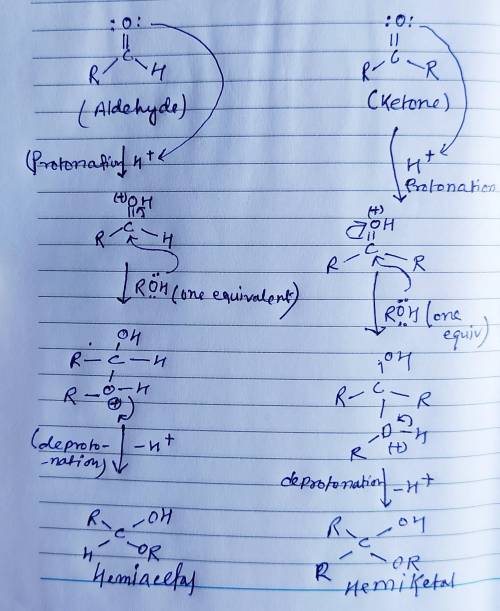
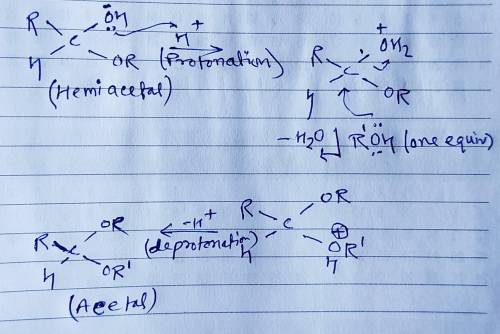
Aldehydes and ketones react reversibly with two equivalents of alcohol in the presence of an acid catalyst to give acetals. Alcohols are poor nucleophiles, and so protonation of the carbonyl oxygen is used to make the carbonyl carbon a stronger electrophile. Addition of the first equivalent of alcohol gives a hemiacetal, a hydroxyether. Addition of the second equivalent of alcohol is accompanied by loss of water to yield the product acetal. Draw curved arrows to show the movement of electrons in this step of the mechanism.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1


Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1
You know the right answer?
Aldehydes and ketones react reversibly with two equivalents of alcohol in the presence of an acid ca...
Questions


Mathematics, 14.03.2020 07:46


Biology, 14.03.2020 07:47


Mathematics, 14.03.2020 07:48





Chemistry, 14.03.2020 07:52



Mathematics, 14.03.2020 07:57

Business, 14.03.2020 07:57

English, 14.03.2020 07:57

History, 14.03.2020 08:00

Computers and Technology, 14.03.2020 08:01

World Languages, 14.03.2020 08:01

Mathematics, 14.03.2020 08:01