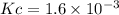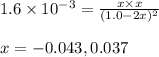Chemistry, 30.03.2020 21:15 4804102262
HI decomposes to H2 and I2 by the following equation: 2HI(g) → H2(g) + I2(g);Kc = 1.6 × 10−3 at 25∘C If 1.0 M HI is placed into a closed container and the reaction is allowed to reach equilibrium at 25∘C, what is the equilibrium concentration of H2 (g)?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
An empty fuel tank can still contain and therefore can be even more dangerous than one full of liquid fuel.
Answers: 1

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1

Chemistry, 22.06.2019 21:50
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2
You know the right answer?
HI decomposes to H2 and I2 by the following equation: 2HI(g) → H2(g) + I2(g);Kc = 1.6 × 10−3 at 25∘C...
Questions

History, 12.10.2021 07:10







Mathematics, 12.10.2021 07:10

Biology, 12.10.2021 07:10

Chemistry, 12.10.2021 07:20


English, 12.10.2021 07:20

Mathematics, 12.10.2021 07:20


Biology, 12.10.2021 07:20


Mathematics, 12.10.2021 07:20

Business, 12.10.2021 07:20

Mathematics, 12.10.2021 07:20

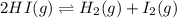
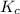 for above equation follows:
for above equation follows:![K_c=\frac{[H_2][I_2]}{[HI]^2}](/tpl/images/0571/2457/ef85e.png)