
Chemistry, 30.03.2020 21:22 usagimiller
Coal gasification can be represented by the equation: 2 C(s) + 2 H2O(g) → CH4(g) + CO2(g) ΔH = ? Use the following information to find ΔH for the reaction above. CO(g) + H2(g) → C(s) + H2O(g) ΔH = -131 kJ CO(g) + H2O(g) → CO2(g) + H2(g) ΔH = -41 kJ CO(g) + 3 H2(g) → CH4(g) + H2O(g) ΔH = -206 kJ

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3

Chemistry, 22.06.2019 22:30
What if it is did darwin used to support his theory of evolution
Answers: 1


Chemistry, 23.06.2019 02:00
Butane gas reacts with oxygen gas to give carbon dioxide gas and water vapor (gas). if you mix butane and oxygen in the correct stoichiometric ratio, and if the total pressure of the mixture is 390 mmhg, what is the pressure (in mmhg) of water vapor after the reaction is completed (temperature and volume do not change).
Answers: 2
You know the right answer?
Coal gasification can be represented by the equation: 2 C(s) + 2 H2O(g) → CH4(g) + CO2(g) ΔH = ? Use...
Questions


History, 16.10.2019 23:00



Business, 16.10.2019 23:00


Computers and Technology, 16.10.2019 23:00





History, 16.10.2019 23:00



Mathematics, 16.10.2019 23:00





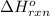 for the reaction is 15 kJ.
for the reaction is 15 kJ.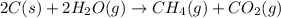
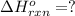
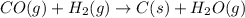
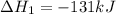 ( × 2)
( × 2)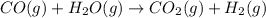
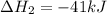
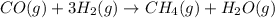
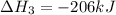
![\Delta H^o_{rxn}=[2\times (-\Delta H_1)]+[1\times \Delta H_2]+[1\times \Delta H_3]](/tpl/images/0571/2803/fd23d.png)
![\Delta H^o_{rxn}=[(2\times -(-131))+(1\times (-41))+(1\times (-206))]=15kJ](/tpl/images/0571/2803/ea721.png)


