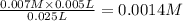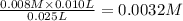A student working in the laboratory prepared the following reactants: 5 mL of 0.007M Cd2+(aq) 10 mL of 0.008M SCN-(aq) 10 mL of 0.5M HNO3(aq) These reagents were mixed and allowed to stand for 10 minutes. The concentration of Cd(SCN)+ in the resulting equilibrium mixture is found to be 5 x 10−4M. Calculate the initial concentration of Cd2+(aq).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Hot air balloons float in the air because of the difference in density between cold and hot air. in this problem, you will estimate the minimum temperature the gas inside the balloon needs to be, for it to take off. to do this, use the following variables and make these assumptions: the combined weight of the pilot basket together with that of the balloon fabric and other equipment is w. the volume of the hot air inside the balloon when it is inflated is v. the absolute temperature of the hot air at the bottom of the balloon is th (where th> tc). the absolute temperature of the cold air outside the balloon is tc and its density is ďc. the balloon is open at the bottom, so that the pressure inside and outside the balloon is the same. as always, treat air as an ideal gas. use g for the magnitude of the acceleration due to gravity.
Answers: 1

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1
You know the right answer?
A student working in the laboratory prepared the following reactants: 5 mL of 0.007M Cd2+(aq) 10 mL...
Questions


History, 05.03.2021 16:40




Mathematics, 05.03.2021 16:40

Mathematics, 05.03.2021 16:40


Mathematics, 05.03.2021 16:40





Mathematics, 05.03.2021 16:40

Mathematics, 05.03.2021 16:40

Health, 05.03.2021 16:40

Mathematics, 05.03.2021 16:40

Mathematics, 05.03.2021 16:40


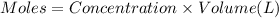
![[Cd^{2+}]=0.007M](/tpl/images/0571/8149/5e85a.png)
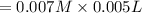
![[SCN^{-}]=0.008 M](/tpl/images/0571/8149/33083.png)
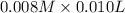
![[HNO_3]=0.5 M](/tpl/images/0571/8149/6e078.png)