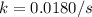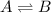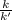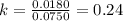Chemistry, 30.03.2020 23:51 eldiamonte
At a given temperature, the elementary reaction A − ⇀ ↽ − B , in the forward direction, is first order in A with a rate constant of 0.0180 s − 1 . The reverse reaction is first order in B and the rate constant is 0.0750 s − 1 . What is the value of the equilibrium constant for the reaction A − ⇀ ↽ − B at this temperature?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2


Chemistry, 22.06.2019 12:30
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3
You know the right answer?
At a given temperature, the elementary reaction A − ⇀ ↽ − B , in the forward direction, is first ord...
Questions



English, 31.08.2020 01:01








Physics, 31.08.2020 01:01





Mathematics, 31.08.2020 01:01

Mathematics, 31.08.2020 01:01


Engineering, 31.08.2020 01:01

Biology, 31.08.2020 01:01