
Chemistry, 31.03.2020 00:14 aubreyfoster
Express the rate of the reaction in terms of the change in concentration of each of the reactants and products.
a. rate=12Δ[HBr]Δt=−Δ[H2]Δt=−Δ[Br2]Δtr ate=12Δ[HBr]Δt=−Δ[H2]Δt=−Δ[Br2]Δt
b. rate=−Δ[HBr]Δt=12Δ[H2]Δt=12Δ[Br2]Δt rate=−Δ[HBr]Δt=12Δ[H2]Δt=12Δ[Br2]Δt
c. rate=−12Δ[HBr]Δt=Δ[H2]Δt=Δ[Br2]Δtra te=−12Δ[HBr]Δt=Δ[H2]Δt=Δ[Br2]Δt
d. rate=Δ[HBr]Δt=−12Δ[H2]Δt=−12Δ[Br2]Δ t

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Why is permeable soil best for plants that need a lot of drainage?
Answers: 1

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1
You know the right answer?
Express the rate of the reaction in terms of the change in concentration of each of the reactants an...
Questions

Mathematics, 03.09.2020 20:01

English, 03.09.2020 20:01

Physics, 03.09.2020 20:01



Mathematics, 03.09.2020 20:01







English, 03.09.2020 20:01







English, 03.09.2020 20:01

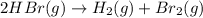
![rate=-\frac{1}{2} \frac{\Delta [HBr]}{\Delta t}=\frac{\Delta [Br_2]}{\Delta t} =\frac{\Delta [H_2]}{\Delta t}](/tpl/images/0572/0368/fc117.png)


