

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 01:00
Which statement characterizes synthetic polymers? a. they come from animals and plants. b. they are found in nature. c. they are made in a lab. d. they are components of starch.
Answers: 1

Chemistry, 23.06.2019 06:00
Give one example of a pure (exact) number and of an estimated (measured) number.
Answers: 2

Chemistry, 23.06.2019 06:40
1.) which of the following is a molecule but not a compound? a.he b.f2 c.h2o d.ch4 2.) what is a physical combination of substances? a.a compound b.a molecule c.a mixture d.an element 3.) what is a chemical combination of substances? a.a compound b.an atom c.a mixture d.an element 4.) what is the relationship between the solute and solvent in a solution? a.they form a compound b.they form a mixture c.they form molecules d.they form chemical bonds 5.) the gases in air dissolve in water. what would be one way to reduce the amount of a gas dissolved in water? a.add more water b.reduce the air pressure c.increase the air pressure d.stir the water 6.) how would you determine the solubility of a substance? a.find how well it dissolved various substances. b.find the mass and the volume of the substance. c.find the temperature at which the substance evaporated. d.find how much i was able to dissolve in a solute. 7.) the periodic table organizes all of the kinds of a.molecules. b.compounds. c.atoms. d.ions. 8.)what distinguishes two substances combined to become a compound vs. two substances combined to become a mixture? a.whether they can be easily separated b.whether they chemically bond together c.whether they both are visible d.whether they are heterogeneous 9.) the principle components of air are: n2 78% o2 21% ar 0.95% co2 0.038% this is a solution of a.molecules and atoms. b.molecules. c.compounds and molecules. d.atoms.
Answers: 1
You know the right answer?
If you combine 250.0 mL 250.0 mL of water at 25.00 ∘ C 25.00 ∘C and 100.0 mL 100.0 mL of water at 95...
Questions

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Biology, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

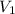 = 250 ml
= 250 ml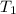 = 25 °c
= 25 °c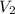 = 100 ml
= 100 ml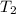 = 95 °c
= 95 °c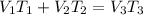
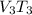
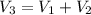
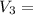 250 + 100 = 350 ml
250 + 100 = 350 ml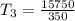
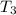 = 45 °c
= 45 °c

