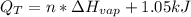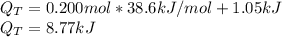Ethanol boils at 78.4 °C with \DeltaΔHvap = 38.6 kJ/mol. A 0.200-mol sample of ethanol is heated from some colder temperature up to 78.4 °C, which requires 1.05 kJ of heat, and then vaporized. What will be the total amount of heat required (for both the heating and the vaporizing)?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3
You know the right answer?
Ethanol boils at 78.4 °C with \DeltaΔHvap = 38.6 kJ/mol. A 0.200-mol sample of ethanol is heated fro...
Questions

Mathematics, 18.03.2021 03:00

Mathematics, 18.03.2021 03:00

Physics, 18.03.2021 03:00

Mathematics, 18.03.2021 03:00

Health, 18.03.2021 03:00

English, 18.03.2021 03:00





English, 18.03.2021 03:00



Mathematics, 18.03.2021 03:00


Chemistry, 18.03.2021 03:00

Mathematics, 18.03.2021 03:00

Mathematics, 18.03.2021 03:00


English, 18.03.2021 03:00