
Chemistry, 01.04.2020 17:42 dilaydi1212
If a system has a reaction quotient of 2.13 ✕ 10−15 at 100°C, what will happen to the concentrations of COBr2, CO, and Br2 as the reaction proceeds to equilibrium?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3

Chemistry, 23.06.2019 03:50
What is the equation fort the alkaline zinc/manganese dioxide cell. a) anode b)cathode c)overall equations.
Answers: 2

Chemistry, 23.06.2019 17:00
Liquid nitrogen is kept at a temperature of -320 degrees. when liquid nitrogen is heated it quickly boils and turns into gas. which pair of pictures represent the change caused by adding heat to liquid nitrogen?
Answers: 3

Chemistry, 23.06.2019 17:30
Hydrogen-2 is also known as deuterium as well as hydrogen-3 is known as tritium hydrogen-1 is our common hydrogen isotope a sample hydrogen gas has 99% hydrogen -1 ,0.8% deuterium , and 0.2% tritium what is the average atomic mass of this mixture of isotope to the thousands place
Answers: 1
You know the right answer?
If a system has a reaction quotient of 2.13 ✕ 10−15 at 100°C, what will happen to the concentrations...
Questions




Mathematics, 24.02.2022 01:10

Computers and Technology, 24.02.2022 01:10


Spanish, 24.02.2022 01:10






Law, 24.02.2022 01:10



History, 24.02.2022 01:10

Mathematics, 24.02.2022 01:10



Computers and Technology, 24.02.2022 01:10

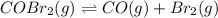
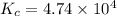
![[COBr_2]=1.58\times 10^{-6}M](/tpl/images/0575/5635/8016e.png)
![[Co]=2.78\times 10^{-3}M](/tpl/images/0575/5635/97ec2.png)
![[Br_2]=2.51\times 10^{-5}M](/tpl/images/0575/5635/9f839.png)
![Q=\frac{[CO][Br_2]}{[COBr_2]}](/tpl/images/0575/5635/e0458.png)
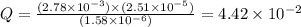
 that means product > reactant. So, the reaction is reactant favored.
that means product > reactant. So, the reaction is reactant favored. that means reactant > product. So, the reaction is product favored.
that means reactant > product. So, the reaction is product favored. that means product = reactant. So, the reaction is in equilibrium.
that means product = reactant. So, the reaction is in equilibrium.

