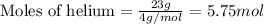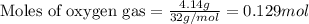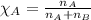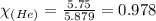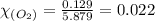Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:40
Water ionizes by the equation h2o(l)⇌h+(aq)+oh−(aq) the extent of the reaction is small in pure water and dilute aqueous solutions. this reaction creates the following relationship between [h+] and [oh−]: kw=[h+][oh−] keep in mind that, like all equilibrium constants, the value of kw changes with temperature.
Answers: 1

Chemistry, 22.06.2019 05:00
Type the letter that represents the correct location for each particle type below.
Answers: 1

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3
You know the right answer?
A 15.0-L scuba diving tank contains a helium-oxygen (heliox) mixture made up of 23.0 g of He and 4.1...
Questions





Advanced Placement (AP), 05.10.2021 04:00

Biology, 05.10.2021 04:00


Mathematics, 05.10.2021 04:00

Mathematics, 05.10.2021 04:00

Social Studies, 05.10.2021 04:00

Mathematics, 05.10.2021 04:00

Social Studies, 05.10.2021 04:00


Mathematics, 05.10.2021 04:00




Computers and Technology, 05.10.2021 04:00


SAT, 05.10.2021 04:00

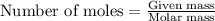 .....(1)
.....(1)