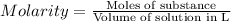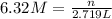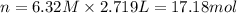Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1
You know the right answer?
How many moles of NaCl are in 2,719 mL of a 6.32 M solution?...
Questions



Advanced Placement (AP), 26.04.2021 23:10


Health, 26.04.2021 23:10

Mathematics, 26.04.2021 23:10

Advanced Placement (AP), 26.04.2021 23:10

English, 26.04.2021 23:10

English, 26.04.2021 23:10

Spanish, 26.04.2021 23:10



Mathematics, 26.04.2021 23:10


Mathematics, 26.04.2021 23:10



English, 26.04.2021 23:10


Mathematics, 26.04.2021 23:10