

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 21:30
What is the correct name for the compound cocl3? a) cobalt(i) chloride b) cobalt(i) chlorate c) cobalt(ii) chlorate d) cobalt(iii) chloride
Answers: 1

You know the right answer?
If 2.87 g of aluminum are reacted with excess copper (II) sulfate and 9.2 g of copper are produced w...
Questions










Biology, 09.10.2019 11:50

Mathematics, 09.10.2019 11:50

History, 09.10.2019 11:50




History, 09.10.2019 11:50



Mathematics, 09.10.2019 11:50

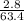
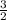 =
=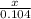
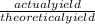 x 100
x 100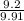 x 100
x 100

