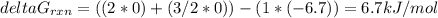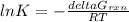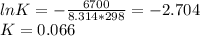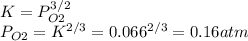Chemistry, 04.04.2020 09:43 emmarieasimon
Consider the decomposition of a metal oxide to its elements, where M represents a generic metal. M 3 O 4 ( s ) − ⇀ ↽ − 3 M ( s ) + 2 O 2 ( g ) What is the standard change in Gibbs energy for the reaction, as written, in the forward direction? Δ G ∘ rxn = kJ/mol What is the equilibrium constant of this reaction, as written, in the forward direction at 298 K? K = What is the equilibrium pressure of O2(g) over M(s) at 298 K? P O 2 = atm

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Motivation cannot be developed with practice; a person either possesses it or they do not.
Answers: 1

Chemistry, 21.06.2019 22:00
If a plot weight (in g) vs. volume (in ml) for a metal gave the equation y= 13.41x and r^2=0.9981 what is the density of the metal?
Answers: 2

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3

Chemistry, 23.06.2019 07:40
Which of the following has expanded our knowledge of the universe beyond our solar system the most? a. manned space travel b. the hubble space telescope c. the pioneer and voyager missions d. the international space station
Answers: 3
You know the right answer?
Consider the decomposition of a metal oxide to its elements, where M represents a generic metal. M 3...
Questions


Mathematics, 05.07.2019 12:40



Biology, 05.07.2019 12:40

English, 05.07.2019 12:40




Chemistry, 05.07.2019 12:40

Mathematics, 05.07.2019 12:40



English, 05.07.2019 12:40

Mathematics, 05.07.2019 12:40



Computers and Technology, 05.07.2019 12:40

Mathematics, 05.07.2019 12:40

History, 05.07.2019 12:40