Chemistry, 04.04.2020 11:34 akatsionis25
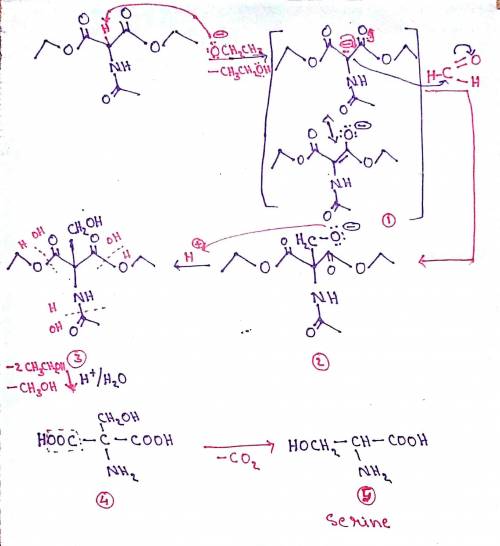
A variation of the acetamidomalonate synthesis can be used to synthesize serine. The process involves the following steps: Ethoxide ion deprotonates diethyl acetamidomalonate, forming enolate anion 1; Enolate anion 1 makes a nucleophilic attack on formaldehyde, forming tetrahedral intermediate 2; Protonation of the oxyanion forms alcohol 3; Acid hydrolysis yields dicarboxyamino alcohol 4; Decarboxylation leads to the final amino acid. Write out the mechanism on a separate sheet of paper, and then draw the structure of dicarboxyamino alcohol 4.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 10:30
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1
You know the right answer?
A variation of the acetamidomalonate synthesis can be used to synthesize serine. The process involve...
Questions






Biology, 24.04.2020 20:13











Mathematics, 24.04.2020 20:13