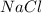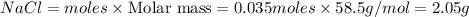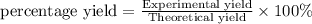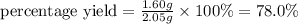Chemistry, 04.04.2020 12:41 shawnaelvaughns
Aqueous hydrochloric acid reacts with solid sodium hydroxide to produce aqueous sodium chloride and liquid water . If of sodium chloride is produced from the reaction of of hydrochloric acid and of sodium hydroxide, calculate the percent yield of sodium chloride. Round your answer to significant figures.

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 04:10
In an experiment, 45g of silicon tetrachloride are treated with 45ml of water. what is the theoretical yield in grams of hcl
Answers: 3

Chemistry, 23.06.2019 12:50
Complete the paragraph to describe the characteristics of a borane molecule (bh3). the lewis structure and table of electronegativities are given. the bond polarities in bh3 are , the molecular shape is , and the molecule is .
Answers: 2

Chemistry, 23.06.2019 13:00
Write the balanced chemical reaction for the formation of fe2(so4)3 from fe2o3 and so3 and determine how many moles of fe2(so4)3 are formed when 12.7 mol of so3 are reacted.
Answers: 1
You know the right answer?
Aqueous hydrochloric acid reacts with solid sodium hydroxide to produce aqueous sodium chloride and...
Questions


Mathematics, 03.08.2019 09:00

Mathematics, 03.08.2019 09:00






Mathematics, 03.08.2019 09:00


Spanish, 03.08.2019 09:00


Biology, 03.08.2019 09:00

Biology, 03.08.2019 09:00


Mathematics, 03.08.2019 09:00

Advanced Placement (AP), 03.08.2019 09:00


Mathematics, 03.08.2019 09:00

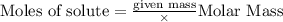
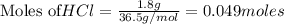
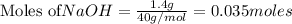
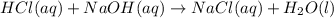
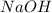 require = 1 mole of
require = 1 mole of 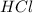
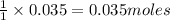 of
of