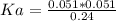Calculate the Ka for the following acid. Determine if it is a strong or weak acid. HClO2(aq) dissolves in aqueous solution to form H+(aq) and ClO2−(aq). At equilibrium, the concentrations of each of the species are as follows: [HClO2]=0.24M [H+]=0.051M [ClO2−]=0.051M

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Which statement describes evidence of a chemical reaction? a) ice melting eliminate b) water boiling c) lighting a match d) grape juice freezing
Answers: 3


Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3
You know the right answer?
Calculate the Ka for the following acid. Determine if it is a strong or weak acid. HClO2(aq) dissolv...
Questions


History, 12.12.2020 16:40


Mathematics, 12.12.2020 16:40

English, 12.12.2020 16:40

Mathematics, 12.12.2020 16:40

Social Studies, 12.12.2020 16:40

Mathematics, 12.12.2020 16:40

English, 12.12.2020 16:40



Mathematics, 12.12.2020 16:40

Physics, 12.12.2020 16:40




Mathematics, 12.12.2020 16:40


Mathematics, 12.12.2020 16:40


![[HClO_2]=0.24M](/tpl/images/0582/3449/64bd9.png)
![[H^+]=0.051M](/tpl/images/0582/3449/e1645.png)
![[ClO_2^-]=0.051M](/tpl/images/0582/3449/4a4bd.png)
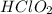 is
is
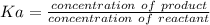
![= \frac{[H^+][ClO_2^-]}{[HClO_2]}](/tpl/images/0582/3449/87292.png)