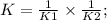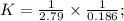Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1


Chemistry, 22.06.2019 19:30
Which one of the following substances would be the most soluble in ccl4? na2so4 h2o ch3ch2ch2ch2oh c4h10 hi
Answers: 1
You know the right answer?
Two reactions and their equilibrium constants are given. A + 2 B − ⇀ ↽ − 2 C K 1 = 2.79 2 C − ⇀ ↽ −...
Questions

English, 25.09.2019 10:30

Mathematics, 25.09.2019 10:30

Mathematics, 25.09.2019 10:30

Physics, 25.09.2019 10:30

Mathematics, 25.09.2019 10:30

Mathematics, 25.09.2019 10:30

Arts, 25.09.2019 10:30

Biology, 25.09.2019 10:30



Mathematics, 25.09.2019 10:30

Mathematics, 25.09.2019 10:30


History, 25.09.2019 10:30





English, 25.09.2019 10:30