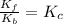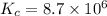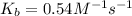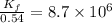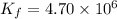Chemistry, 04.04.2020 14:31 eburnhisel2023
At 700 K the equilibrium constant KC for the reaction between NO(g) and O2(g) forming NO2(g) is 8.7 × 106. The rate constant for the reverse reaction at this temperature is 0.54 M–1s–1. What is the value of the rate constant for the forward reaction at 700 K?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

You know the right answer?
At 700 K the equilibrium constant KC for the reaction between NO(g) and O2(g) forming NO2(g) is 8.7...
Questions

History, 25.05.2021 17:10



Mathematics, 25.05.2021 17:10

Spanish, 25.05.2021 17:10

Social Studies, 25.05.2021 17:10

English, 25.05.2021 17:10

Mathematics, 25.05.2021 17:10

English, 25.05.2021 17:10




Mathematics, 25.05.2021 17:10

Computers and Technology, 25.05.2021 17:10






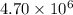
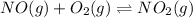
![K_c=\frac{[NO_2]}{[NO][O_2]}](/tpl/images/0582/5120/67e8f.png)
![R_f=K_f[NO][O_2]](/tpl/images/0582/5120/9cd84.png)
![R_b=K_b[NO_2]](/tpl/images/0582/5120/d91b1.png)
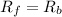
![K_f[NO][O_2]=K_b[NO_2]](/tpl/images/0582/5120/7432e.png)
![\frac{K_f}{K_b}=\frac{[NO_2]}{[NO][O_2]}](/tpl/images/0582/5120/419f2.png)