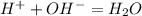Under what condition is the OH^- ion concentration in water expected to be zero?
A. In a...

Chemistry, 05.04.2020 05:16 SMURFETTE86
Under what condition is the OH^- ion concentration in water expected to be zero?
A. In a solution of strong acid
B. In a solution of strong base
C. In a solution of weak base
D. never

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2


Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
Questions

Computers and Technology, 13.12.2019 01:31



















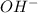 ion is also referred to as Hydroxide ion.
ion is also referred to as Hydroxide ion.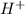 (hydrogen) ion.
(hydrogen) ion.