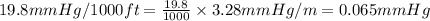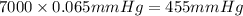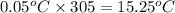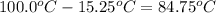The vapor pressure of a substance describes how readily molecules at the surface of the substance enter the gaseous phase. At the boiling point of a liquid, the liquid's vapor pressure is equal to or greater than the atmospheric pressure exerted on the surface of the liquid. Since the atmospheric pressure at higher elevations is lower than at sea level, the boiling point of water decreases as the elevation increases. The atmospheric pressure at sea level is 760 mmHg. This pressure decreases by 19.8 mmHg for every 1000-ft increase in elevation. Elevation Pressure0 m 760 mmHg1000 m 695 mmHg2000 m 630 mmHgThe boiling point of water decreases 0.05 ?C for every 1 mmHg drop in atmospheric pressure. A) What is the boiling point of water at an elevation of 7000m ?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1
You know the right answer?
The vapor pressure of a substance describes how readily molecules at the surface of the substance en...
Questions













Chemistry, 29.08.2020 14:01






English, 29.08.2020 14:01

Chemistry, 29.08.2020 14:01

 meter
meter