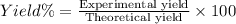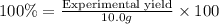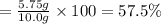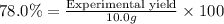Chemistry, 06.04.2020 20:18 Thejollyhellhound20
Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+):
CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3C H2CH2CO2CH2CH3(l)+H2O(l)
Part A
Given 7.60 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield?
Express your answer in grams to three significant figures.
Part B
A chemist ran the reaction and obtained 5.75 g of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.60 g of butanoic acid and excess ethanol?
Express your answer in grams to three significant figures.
Part C
The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.60 g of butanoic acid and excess ethanol?
Express your answer in grams to three significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 21:40
Tooth enamel consists mainly of the mineral calcium hydroxyapatite, ca_10(po_4)_6(oh)_2. trace elements in teeth of archaeological specimens provide anthropologist with clues about diet and diseases of ancient people. students at hamline university measured strontium in enamel from extracted wisdom teeth by atomic absorption spectroscopy. solutions with a constant total volume of 10.0 ml contained 0.726 mg of dissolved tooth enamel plus variable concentrations of added sr. added sr find the concentration of sr in the 10 ml sample solution in parts per billion = ng/ml. find the concentration of sr in tooth enamel in parts per million = mu g/g.
Answers: 2

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 22.06.2019 23:00
What is the measured amount of a product obtained from a chemical reaction?
Answers: 1
You know the right answer?
Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry...
Questions



Mathematics, 12.12.2020 15:50

History, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50

Social Studies, 12.12.2020 15:50

English, 12.12.2020 15:50

Chemistry, 12.12.2020 15:50

Mathematics, 12.12.2020 15:50


Mathematics, 12.12.2020 15:50

History, 12.12.2020 15:50


Mathematics, 12.12.2020 15:50





Mathematics, 12.12.2020 15:50

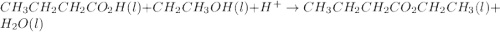
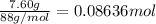
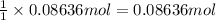 of ethyl butyrate
of ethyl butyrate