
Chemistry, 07.04.2020 02:35 uglyturtle15
Consider the reaction of magnesium metal with hydrochloric acid to produce magnesium chloride and hydrogen gas. If 3.29 mol of magnesium and 3.29 mol of hydrochloric acid are reacted, how many moles of excess reactant are left over?
a. 6.58 mol
b. 3.29 mol
c. 2.47 mol
d. 40.0 mol

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:50
Which real-world scenarios below represent physical and chemical changes? -running a car -exploding fireworks -mixing water and powdered drink mix -combining oil and vinegar to make salad dressing -taking aspirin for a headache -diluting bleach with water-digesting dinner-spreading peanut butter on bread
Answers: 2

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 02:30
Which piece of equipment would me most useful for measuring the volume of some water? a. pan balance b. graduated cylinder c. tweezers d. flask quick
Answers: 2
You know the right answer?
Consider the reaction of magnesium metal with hydrochloric acid to produce magnesium chloride and hy...
Questions




Mathematics, 24.03.2020 23:55



English, 24.03.2020 23:55


History, 24.03.2020 23:55

Mathematics, 24.03.2020 23:55



Computers and Technology, 24.03.2020 23:55


Business, 24.03.2020 23:55

Social Studies, 24.03.2020 23:55

History, 24.03.2020 23:55

Mathematics, 24.03.2020 23:55


History, 24.03.2020 23:55

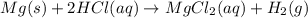
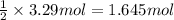 of magnesium metal
of magnesium metal

