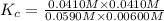Chemistry, 07.04.2020 03:15 sethlynn2003
Calculate the equilibrium constant for the reaction using the balanced chemical equation and the concentrations of the substances at equilibrium.
Use the
appropriate significant figures in reporting the answers.
CO(g) + H2O(g) ⇌ CO2(g) + H2(g) [CO] = 0.0590 M; [H2O] = 0.00600 M;
[CO2] = 0.0410 M; [H2] = 0.0410 M
K =-

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
How to solve 4 nh3(g) + 5 o2(g) > 4 no(g) + 6 h2o(g) in chemistry
Answers: 1

Chemistry, 22.06.2019 11:00
The twister and runaway train are two coasters at the same amusement park. both coasters start at the same height. the coaster for the twister is twice the mass of the coaster for the runaway train. which roller coaster has greater gravitational potential energy at the start of the ride?
Answers: 1

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
You know the right answer?
Calculate the equilibrium constant for the reaction using the balanced chemical equation and the con...
Questions

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

Mathematics, 16.09.2020 01:01

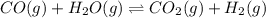
![[CO]=0.0590 M,[H_2O]=0.00600 M](/tpl/images/0585/8718/63397.png)
![[CO_2]=0.0410 M,[H_2]=0.0410 M](/tpl/images/0585/8718/efcda.png)
![K_c=\frac{[CO_2][H_2]}{[CO][H_2O]}](/tpl/images/0585/8718/fbbde.png)