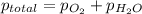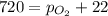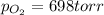4. A 250 mL sample of oxygen gas is collected by water displacement. As a result, the oxygen is saturated with water vapor. The partial pressure of water vapor at the prevailing temperature is 22 torr. Calculate the partial pressure of the oxygen if the total pressure of the sample is 720 torr.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2


Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3
You know the right answer?
4. A 250 mL sample of oxygen gas is collected by water displacement. As a result, the oxygen is satu...
Questions






Chemistry, 07.09.2020 03:01

History, 07.09.2020 03:01


Health, 07.09.2020 03:01


Mathematics, 07.09.2020 03:01

History, 07.09.2020 03:01



English, 07.09.2020 03:01

Biology, 07.09.2020 03:01



Physics, 07.09.2020 03:01

Biology, 07.09.2020 03:01

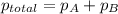
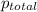 =total pressure of gases = 720 torr
=total pressure of gases = 720 torr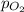 = partial pressure of
= partial pressure of 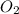 = ?
= ?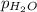 = partial pressure of
= partial pressure of 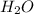 = 22 torr
= 22 torr